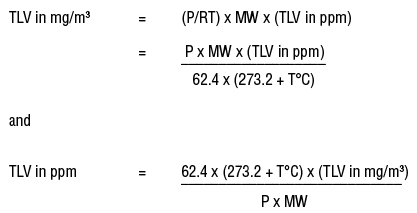
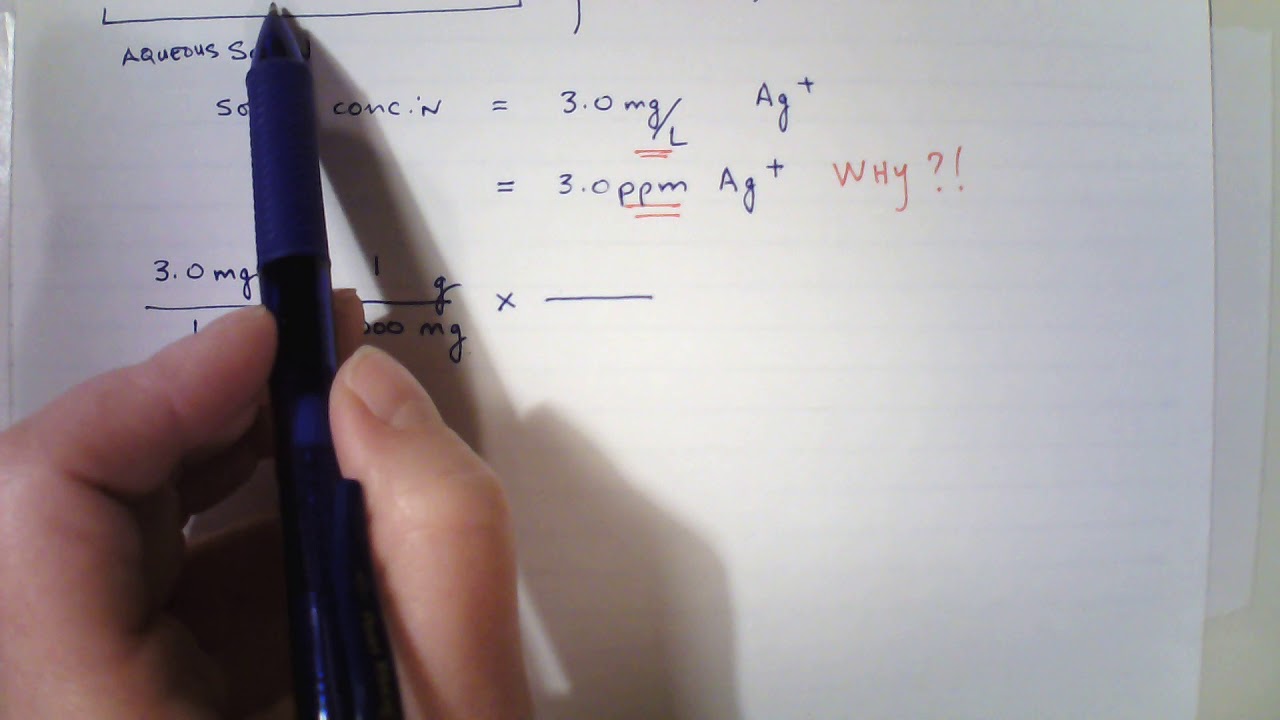Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download

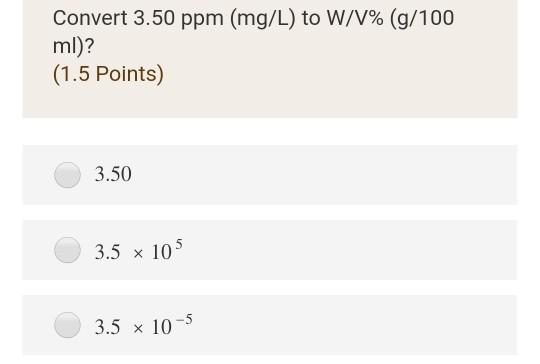
PPT - mg/L - milligrams per litre (10-3 g per litre) = mg per 1,000,000mg THUS = ppm PowerPoint Presentation - ID:4270418

Concentrations expressed as mg L -1 (ppm) by labs must be divided by... | Download Scientific Diagram

10.30 mg of O2 is dissolved into a liter of sea water of density 1.03 g/mL . The concentration of O2 in ppm is .

SOLVED: 32 Using the following equation, determine the average concentration (moles per liter) of Ca2+ ions present in your water: Record the concentration in Data Table 2. mol Ca2+ mol EDTA 1